Laboratory of Bioengineering and Nanosciences (LBN)


The LBN (Laboratory for Bioengineering and Nanoscience) was established in 2007. The LBN is known for combining specific inorganic scaffolds (such as porous silicon) with innovative microscopy techniques—including confocal Raman and multiphoton microscopy—for tissue engineering using human dental pulp stem cells (DPSCs) and induced pluripotent stem cells (iPSCs).
The LBN is located at the heart of the Faculty of Dentistry in Montpellier and is directed by Professor Frédéric CUISINIER. The LBN is part of the Department of Health Biology (DBS).
LBN is ISO 9001 certified for its public research processes, and the EDMOS platform is currently undergoing certification for its private research processes.
Since 2021, the LBN has been home to three interdisciplinary research groups and a technological platform.
The EDMOS platform is a collaborative platform that brings together technical resources from LBN, Institut Mines Telecom Ales, and CHU Montpellier.
The researchers are affiliated with the School of Dentistry, the School of Pharmacy, and the Department of Chemistry in the School of Science.
GROUP 1
PIERRE-YVES COLLART DUTILLEUL (GROUP LEADER)
- Valérie ORTI (Associate Professor)
- Stéphane BARTHELEMI (Associate Professor)
- Ivan PANAYOTOV (MCU PH)
- Jean Claude EGEA (Master of Clinical Sciences in Public Health)
- Habib BELAID (Associate Professor)
- Jerome PRADOS (Assistant Professor)
- Marc ZINDEL (Technician)
TASKS
- Porous silicon for the proliferation and differentiation of dental pulp stem cells
- PEEK functionalization for cell adhesion and mineralized tissue formation
- Bifunctional peptide for epithelial adhesion to titanium
- Bone and cartilage formation monitored by Raman spectroscopy
- DPSC for anticancer drug delivery
- DPSC Conditioned Medium for Cell and Tissue Regeneration
GROUP 2
AZEL ZINE (PU, Group Leader)
GROUP MEMBERS
- Azel ZINE (PU, Group Leader)
- Veronique MONTERO (MCU)
- Olivier ROMIEU (Master of Clinical Sciences, Associate Professor)
- Bayan AASAR (Ph.D. student)
- Meryam ZAROUKI (Research Technician)
- Maëlis LUCAS (Project Manager)
- Damien VERET (Postdoctoral Researcher)
Stem Cells, Organoids, and Otic Neurosensory Regeneration
Sensorineural hearing loss is often caused by the degeneration of spiral ganglion neurons (SGN) in the inner ear, which relay auditory signals to the brain. These SGN do not regenerate after degeneration. Dental pulp stem cells (DPSCs) are neural crest-derived ectomesenchymal stem cells, similar to cranial neural crest cells (NCC). These NCC partially contribute to the formation of SGN during inner ear development. DPSCs are promising candidates for cell therapy because they possess migratory properties, enabling them to migrate after transplantation; they can differentiate into sensory neurons and glial cells; and they can be harvested in relatively large numbers. We hypothesize that human DPSCs can be used for cell-based regeneration and repair of SGNs.
Project 1: Production of human SGN from DPSCs and iPSCs using 2D and 3D cultures
The main goal of this task is to differentiate and characterize human otic neurosensory progenitors and SGN using a cellular platform for the in vitro production of differentiated cell types from either iPSCs or DPSCs. The stepwise guided differentiation of stem cells into SGN is not straightforward, since there is no single marker that can be used to distinguish this specific cell type. However, by using high-throughput RNA-seq gene expression analysis, it will be possible to more thoroughly determine whether cohorts of known otic neurosensory progenitors and SGN markers follow a sequential pattern over the course of in vitro differentiation. We will compare the overall differentiation trajectory toward the human otic neurosensory lineage in both DPSC- and iPSC-derived otic neuronal progenitors and SGN. Identity and purity control experiments for otic neuroprogenitors and their neurosensory trajectories will be conducted using confocal Raman spectroscopy in collaboration with Team 3 and the Edmos Platform.
Project 2: Potential of sugar-based angiogenic agents for vascularization of organoids during iPSC and DPSC differentiation into SGN neurosensory cells
One of the unique aspects of organoid models is their ability to resemble, to some extent, the in vivo organ or tissue from which they were derived, and to facilitate relevant cell-cell and cell-matrix interactions. One of the key limitations in using organoid approaches to generate tissues is that upon reaching a certain size, organoids cease to proliferate and develop a necrotic core. To maintain the complexity of organoids, it is necessary to prevent the formation of the necrotic inner core, which leads to premature differentiation in the outer layers of organoids. This phenomenon, in turn, has largely been attributed to a lack of organoid vascularization. We will investigate a panel of angiogenic effectors derived from Mannose-6-phosphate (M6P) on the neurodifferentiation of either DPSCs or iPSCs, and on the development of organoid vascularization
Project 3: Effects of Transplanting Neurosensory Progenitors in Models of SGN Degeneration To test the regenerative and synaptogenic capabilities of DPSC/iPSC-derived otic neurosensory progenitors following engraftment, we will first usean in vitro co-culturemodel with denervated cochlear organotypic explants. We have previously demonstrated that hiPSC-derived otic progenitors are able to engraft and differentiate in the damaged cochlea (Lopez et al., 2019).
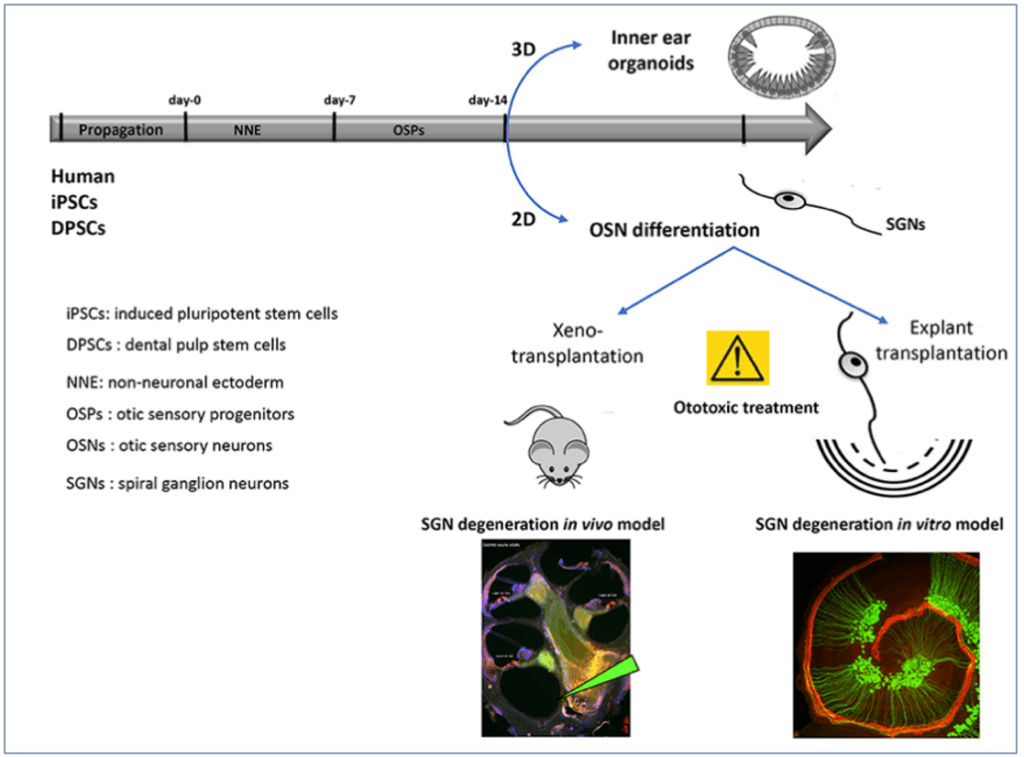
General overview of in vitro differentiation of auditory neurons and engraftment in SGN degeneration models
COLLABORATIONS:
- Albert Edge (Harvard Medical School)
- John Devos (IRMB, CHU, Montpellier)
- Christian Chabbert (CNRS, AMU Marseille)
- Pierre Gaudriault (Cherry Biotech, Rennes)
- Annelies Schrott-Fischer (Medical University of Innsbruck, Austria)
GROUP 3
AMEL SLIMANI (GROUP LEADER)
- Frédéric CUISINIER (Associate Professor)
- Jean Valcarcel (Associate Professor)
- Michel Fages (Full Professor)
- Ivan Panayotov (MCU-PH)
- Jean Cédric Durand (Associate Professor)
- Alban Desoutter (Technical Assistant, LBN)
- Elias Estephan (Associate Professor)
Our scientific objective is to identify new photonic biomarkers for oral diseases, primarily dental caries, periodontal diseases, and oral cancer. These conditions will be studied using various spectroscopic methods and devices. We have organized this project around these methods, although they can be combined to increase the specificity and sensitivity of the biomarkers.
1. 3D intraoral scanners (IOS):
Oral cancer is a significant health problem that is often diagnosed at a late stage. Oral cancers are characterized by changes in tissue color as well as thickening of the oral mucosa. With regard to tissue thickening, creating a 3D mesh of the oral cavity is a valuable tool for developing standardized biomarkers of cancerous lesions. Autofluorescence, color, and 3D meshes could be combined to detect oral cancer. We will focus our work on exploring the use of these two imaging modalities to optimize cancer detection. To this end, 3D meshes of malignant lesions and healthy tissue will be collected. Segmentation and color spectral analysis will be used to identify biomarkers through multivariate statistical analysis. This approach can be extended to periodontal disease.
2. BCARS for the detection of oral cancer
The diagnosis of malignant and cancerous lesions in the oral cavity is the primary focus of the development of new imaging techniques. Malignant tissues undergo biochemical changes known as tumor markers, which cause variations in the vibrational spectra. Raman spectra from diseased and healthy tissue are compared for diagnostic purposes. Raman biomarkers complement biopsy and provide real-time diagnosis. The first part of the task is to compare existing standard biopsy analysis with our label-free B-CARS signal. The relevant data analysis could detect changes in tissue, with the potential for use in the operating room.
3. Early detection of caries lesions using Brillouin/Raman biomarkers
Early-stage caries lesions are difficult for dentists to detect, and even more so in teledentistry. In our laboratory, we have developed aWSL in vitro modeland used confocal Raman spectroscopy and multiphoton microscopy images to evaluate structural changes in white spot lesions. The main objective of this project is to adapt a new non-invasive biomedical imaging technology based on Brillouin light scattering to measure the biomechanical properties of hard tissues and biomaterials. Molecular analysis of dental tissues using Raman spectroscopy is being employed to characterize the mineral phase in synthetic compounds and natural tissues. Our project involves using confocal Raman microscopy coupled with a home-built Brillouin spectrometer. The first step will be to demonstrate the feasibility of simultaneous biomechanical and biochemical imaging to obtain photonic biomarkers for the early diagnosis of caries and WSL by studying in vitro incipient caries lesions and animal-induced periodontal lesions. Comparison with healthy samples will enable statistical analysis to identify a biomarker for detecting caries at an early stage, which is the final step.
Partnerships:
1- L2C-UMR5221 CNRS: We will continue our strong collaborations through our involvement in the funding and construction of the BCARS (Nanobiophonic team) and the construction of the Brillouin Spectrometer (B. Rufflé, D. Felbacq, Physics of the Glassy State team). We will develop a partnership for colorimetry in teledentistry (F. Geniet, Complex Systems and Nonlinear Phenomena team).
2- LIRMM: All metrology-related work for IOS is discussed and carried out in collaboration with LIRMM (G. Subsol, ICAR team).
3- LNE: Regarding the metrology of IOS, the characterization of objects, and the samples tested, we collaborate with a team from LNE (Laboratoire National de Métrologie et d’Essai, Nîmes).
EDMOS PLATFORM

EVALUATION OF MEDICAL DEVICES IN DENTISTRY AND STOMATOLOGYOGY

The EDMOS platform is a consortium of laboratories and research centers united by a common goal: the design, innovation, and development of dental medical devices.
EDMOS consists of three main structures:
- LBN Laboratory
- C2MA Laboratory
- Montpellier University Hospital Dental Clinic
Our Objectives:To establish a partnership between the private sector and leading academic researchers. To provide our technical platforms to companies interested in developing dental medical devices.
Projects and contracts with the EDMOS platform include the preparation of patents and scientific publications based on processed and validated data to provide scientific validation of your device or product.
MORE INFORMATION: HTTPS://EDMOS.EDU.UMONTPELLIER.FR/
